Introduction
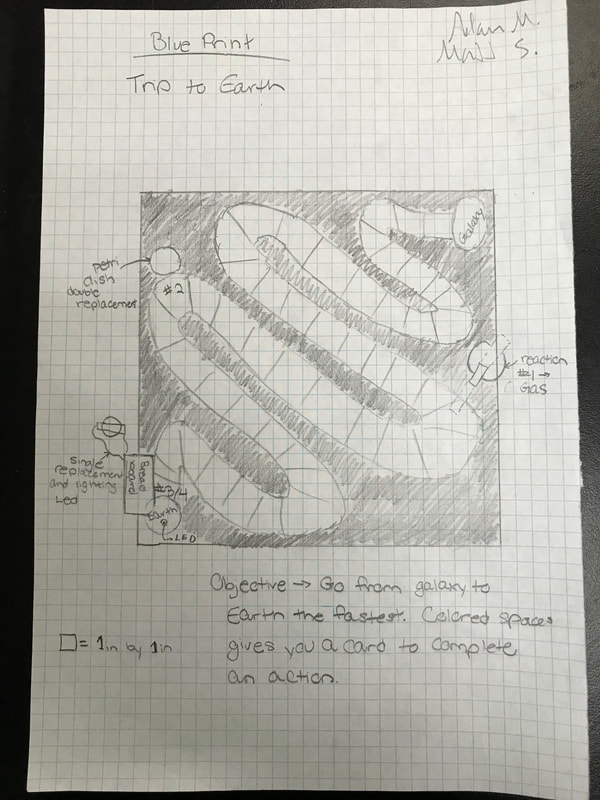
The Chemical Dominoes project was a project to learn about chemical reactions through games. The goal of the project was to find a fun way to showcase and learn about chemical reactions. Certain criteria needed to be met in each game, including a single replacement, double replacement, production of gas, and LED lighting reaction. My group decided to make a space themed game which we called "Trip to Earth." The goal of the game was to be the first alien to reach Earth, starting from your home galaxy. We chose to target a younger audience, and so colored the game with vibrant colors, along with designing a less skill intensive play through so all ages could enjoy it. The game had players choose a spaceship as their game pice, and then roll a die to advance through the board. Along the board were green and red spaces. If a player landed on one of these, they picked up a card from the correspondingly colored pile and followed its instructions. There were also spaces marked as "Reaction Spaces." If a player were to land on one of these, they were to follow the instructions given in the "Reactions" section of the rules.
Trip to Earth |
Chemical ReactionsSingle Displacement Reaction
A chemical reaction by which one element replaces another element in a compound. It can be represented as: A + B-C → A-C + B. Chemical Equation : Al + CuCl2 → Cu + AlCl3 (Aluminum and Copper Chloride yields Copper and Aluminum Chloride) In my groups project, the single replacement reaction was used to cut a circuit. This was done by connecting two wires by a strip of aluminum foil, and then dipping the foil into copper chloride. Since aluminum is more reactive than copper, it replaced the copper in the aqueous solution. The strip then fell apart, cutting off the flow of electricity through the circuit. This then made the electricity flow through a different circuit, which including an LED light, thus lighting up the bulb. The LED was turned on when a player reached Earth, winning the game. It signified the humans lighting a massive beacon to hopefully attract more aliens to come and visit. Double Displacement Reaction A chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products. It can be represented as: AB + CD → CB + AD. Chemical Equation : CuCl2 + K2CrO4 → K2Cl2 + CuCrO4 (Copper Chloride and Potassium Chromate yields Potassium Chloride and Copper Chromate) We used the double replacement reaction to penalize players that were unlucky enough to land on it. The compounds formed from the reaction had a rust color, so we made the player skip a turn in order to repair the rusted metal of their spaceship. Gas Production Reaction A chemical reaction where gas is a product. Chemical Equation : NaCHO3 + CH3COOH → CO2 + H2O + Na + CH3OO (Sodium Bicarbonate and Acetic Acid yields Carbon Dioxide, Water, Sodium, and acetate) For our board game, a gas producing reaction was incorporated to resemble a wormhole. On our board was a lever connecting two parts of the game. This represented a wormhole players could use to warp further into the game. To activate it, players had to land on the corresponding reaction square. Then, they mixed baking soda (sodium bicarbonate) and vinegar (acetic acid) together in a flask with a balloon wrapped over the tip. The baking soda and vinegar reacted, which resulted in the formation of carbon dioxide. This then filled up the balloon on top of the flask, and then tipped the lever to symbolize traveling through the wormhole. LED Reaction We also had to include a reaction which lit up an LED. To do this, as described above, we used a single replacement reaction to switch from a circuit passing through an aluminum strip to one that lit up the LED. |
Reflection
Overall, I felt that the project went well. My group was able to successfully create and present a board game that educated the players on chemical reactions. I believe I did well as a task manager. Throughout our work time, I was constantly dividing up the work we needed to do, and designating it to my group members in order to make sure everyone was working and we were being efficient. Another peak I believe I had was actually presenting the game. During both my freshmen and sophomore years at high school, I hated presentations because of the public speaking involved. For this presentation however, I prepared by rehearsing my speaking lines and made a notecard in case I forgot a line. This helped calm me, as I knew if I forgot a specific equation I could just look at my notecard. As a result, I presented way better than I had in my previous years.
A place where I struggled was staying on task. Although I gave out tasks to my project, I sometimes struggled to complete mine as I got distracted from other groups or my friends. Although I still got my work finished, it would have been more beneficial to keep focus in order to get more work done. Another pit of mine was empathy. I definitely could have collaborated more with some of my group members to get them more involved in our project. Not being empathetic hurt our group because it prevented us from working well with each other at times.
If I were to redo this project, I would focus on the visual aspects of our game, to make it look more appealing to play. I would also revise the integration of our chemical reactions, as sometimes we had to stop the players in order to run the reactions and explain what they did.
A place where I struggled was staying on task. Although I gave out tasks to my project, I sometimes struggled to complete mine as I got distracted from other groups or my friends. Although I still got my work finished, it would have been more beneficial to keep focus in order to get more work done. Another pit of mine was empathy. I definitely could have collaborated more with some of my group members to get them more involved in our project. Not being empathetic hurt our group because it prevented us from working well with each other at times.
If I were to redo this project, I would focus on the visual aspects of our game, to make it look more appealing to play. I would also revise the integration of our chemical reactions, as sometimes we had to stop the players in order to run the reactions and explain what they did.